- Blog
- Hitfilm pro vs studio
- Film indonesia asmara dua diana
- Avi joiner 2-03 registration key
- Hong kong tourism board india
- Coolio gangsters paradise full album zip
- Bass single coil vs humbucker
- Real action pose collection 04 scans
- I doser ecstasy
- Did little master season 2 7th july 2012
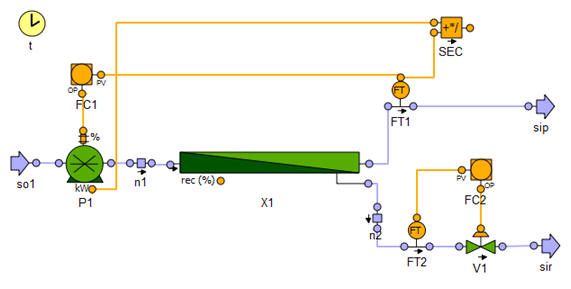
- Reverse osmosis pro ii simulation
- Lifan pony 100 review
- Behringer x32 producer manual
- Ajith spb
- Mirai nikki manga reader
- Integrity air 3000 reviews
- National academies press juvenile delinquency
- Cities skylines how to change road direction
- Propresenter 7 upgrade cost
- Zebra designer pro 2 activation key
- Metalix cnckad download
These calculated permeability coefficients along with the above-mentioned correlations are used to estimate the permeability coefficients of other species present. The user's actual test condition data are the key information for calculating the permeability coefficients of water and the test solute for the membrane element. User-entered test conditions data can then be entered for this membrane data. In addition to physical dimensions (i.e., membrane area), the product sheets also report performance of the membrane (i.e., permeate flux, recovery and rejection percentage for NaCl or other solutes) at specific test conditions. For other neutrals, self-diffusivities relative to water have been used to correlate the permeability.įor information on a particular membrane, the RO block relies on the data from commercial membrane manufacturers who provide a public product specification sheet (available on the Internet) for each type of membrane. For some organics with same homologous group (i.e., alcohols, phenols, acids), the rejections have been correlated by calculating the topological parameters characterizing molecular structure.
The permeability of cations and anions calculated as a function of hydration numbers follow the typical trends. In general, the typical order of rejection of cations by reverse osmosis membranes follow Fe 3+ > Ni 2+ Cu 2+ > Mg 2+ > Ca 2+ > Na+ > K+, and PO43- > SO42- > HCO 3- > Br- > Cl- > NO 3- F- for anions.
In the OLI reverse osmosis membrane model, permeability of ions has been correlated with their hydration numbers. Regardless of membrane type, or the type of experiment, or the membrane configuration, the salt permeabilities are inversely proportional to the hydrated radii of the ions. Thus, the real radius of an ion, that is, the hydrated ion radius, rather than the absolute ionic crystal radius. However, in a water-based environment, a different number of water molecules surrounds each cation and anion. This is due to the sieving actions of the membranes. Molecular size of the ions strongly affects the transport characteristics of the ions. The solute flux through the membrane is a function of the solute permeability constant and the solute concentration gradient across the membrane. The water flux through the membrane is a function of the water permeability coefficient, the hydrodynamic pressure difference and osmotic pressure across the membrane. Following this approach, the model calculates the transfer of ions and water through polymeric membranes via a solution diffusion mechanism, because of dissolution of permeates in the membrane materials. OLI used a solution-diffusion approach to model the transport mechanism inside the membrane.
